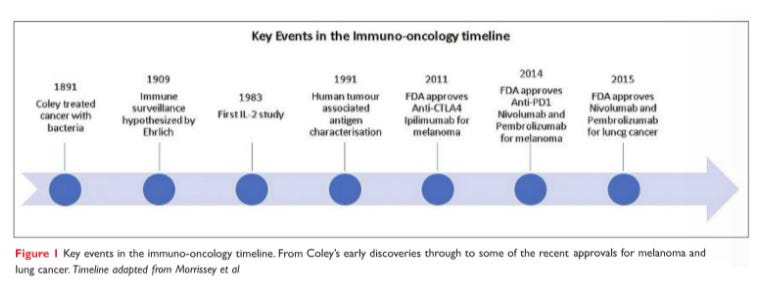
Earlier this year, President Joe biden made a passionate plea to revive the famous American tradition of fighting a ‘war on cancer’ or creating a ‘cancer moonshot’. This adage goes back to more than 50-years ago, when chemotherapy was first introduced.
But are we closer to winning this war?
This essay will serve as a deep dive into this topic with some insight into what the next 20 years may hold. I hope this will be helpful and a thorough review for all, regardless of familiarity with the topic.
At its core, the challenge of curing cancer is synonymous with the challenge of fighting evolution itself. Cancer cells can be considered the next step of an iterative process of cellular evolution, they hack our intrinsic physiology to grow faster and more efficiently than their predecessors. They do this in a way that also evades our body’s defences (immune system) can find and destroy them.
Furthermore, if they are ever discovered by the host, these abnormal cells often develop the ability to evade destruction through swift rescue pathways. This is the failure mode that is most common when cancer goes metastatic or when drugs that worked well initially stop working entirely (The latter is known as drug resistance).
In the 50s and 60s when the first chemotherapy drugs hit the market, there was hope of things getting better for cancer patients, but increasing drug resistance and the high burden of toxicity of these treatments made it clear that more precise and effective firepower was needed. We will review all the main treatment modalities one-by-one and what the biggest impact on everyday patients has come from.
1. Chemotherapy.
This “war’s” inception came in the late 1940s after Sydney Farber used a folate antagonist, aminopterin, to block the function of folic acid (needed for DNA synthesis) in patients with leukemia. Realizing its effectiveness, he published what was the foundation piece of research for modern chemotherapy. On the shoulders of his gigantic contributions, researchers have now made great strides in understanding the pathophysiology of cancer and developing new effective treatments. These are both the oldest and now most widely used type of treatment for cancer.
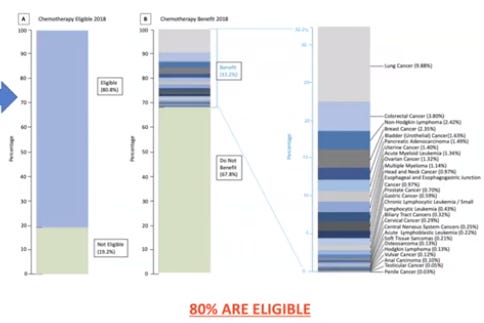
Chemotherapy drugs are cytotoxic, they kill cancer cells using a varitety of mechanisms, but as that definition suggests, they also affect normal cells in the body. This causes gruesome side effects to patients undergoing chemotherapy and significant toxicity overall. Today, the most commonly used chemotherapy drugs are platinum-derived alkylating compounds such as cisplatin and carboplatin. All-in-all Chemotherapy is still the backbone of cancer care in 2022 with a median response of 50% across the board and over 80% of patients still being elegible for some type of chemotherapy drug.
It is still exceedingly rare that any cancer patient in the U.S. today is not exposed to this drug class at one point or another in their treatment course. And for good reasons, these drugs work well and achieve their goal. Unfortunately, they also carry a ton of side effects such as (but not limited to) hair loss, nausea, vomiting, diarrhea and sometimes even infertility and organ damage.
This “ugly side” of chemotherapy drugs and the fact that half of all patients still did not respond is the reason precision oncology was such a wide area of interest in the past 30 years. Many believed that precision medicine would deliver a cancer-specific drug that could cure cancer without harming patients (this idea of a “single bullet” for cancer). And from this dream came an incredible amount of research funding which led to advances in the fields of radiation and surgical oncology, immunotherapy, and genomics.
But how far have we really come with these advances, what is the verdict?
I will split this answer into two parts, starting with research advances that were touted as groundbreaking (early-phase trials) but did not deliver at the bedside (phase 4 and beyond).
But before I get there, there is one last piece of context that is key to understanding why some of these drugs have performed the way they did. Once a patient is advanced enough in their cancer (metastatic phase), very few things can actally help overcome the incoming collapse. The body is overrun by malignant cells in every organ that hijack our biochemical processes for the sole benefit of the invador. Most of the time, failure to thrive and multi-organ failure is the unfortunate next step. In a specific set of cancers, we have created magic bullets that can precisely strike in every part of the body where cancer has spread to undo this scary process, but alas for most people today, such hope is still not accessible.
In fact, the difference between the outcomes of patients with stage 1 to stage 2 cancers versus their stage 4 counterparts (metastatic) is so staggering that it almost should be considered a different disease entirely. One of the oncology attendings that I worked with told me it was sometimes to explain it to patients and their families in terms of “tortoises and alligators”. An early-stage cancer is a tortoise, it’s not great to have but it’s also not deadly and it might not affect life expectancy overall. But stage 4 cancer quickly becomes an alligator, and more often than not, it shortens life expectancy.
As far as alligators go, Pancreatic cancer is one of the worst of them all. This is not a “war” you ever want to be fighting. Such language of “fighting” is disrespectful to all the patients every year who suffer through tratements early (think 54% response rate) and still face these insurmountable odds at the end of that.
As you can see here, around 95% of patients don’t make it past the first year after metastatic diagnosis. This trend is seen in many other advanced cancers but it is important to review the literature on a case-by-case basis.
Part 1 - The underperformers
This includes 71 drugs of the last FDA-approved cancer drugs (x-axis) plotted against their survival rates (PFS and OS) after treatment (y-axis). These drugs were advertised as “moonshots” or maybe “game-changers” during the research and development pipeline but as you can see above, they have not delivered on these promises with average survival for someone experiencing “an alligator” counted in months, not years.
In other studies, these results have been replicated. For stage-4 solid tumors at least, the average life expectancy of patients, even with the best treatment available, is usually between 6 months (at worst) to 2 years (optimistic prognosis).
For example, the drug Sorafenib which generated a lot of hype in the world of hepatocellular carcinoma has simply not held up in later stages of trials. Probably because of the differences between patients eligible for trials, they might be a different demographic from patients being treated in “the real world”.
This is simply not good enough given the investment in the field and has been extremely humbling for me. This is also why many new trainess shy from the field of oncology, they viscerally experience this on the wards and realize that many patients simply don’t make it.
What about Immunotherapy?
Our immune system is the body's best natural defence against outside entities. Cancer cells because of the changes they undergo also have a foreign signature compared to the cells they arise from. These two principles can be combined to design powerful precision drugs that enlist the immune system to do all the dirty work and thus have less side-effects than classic cytotoxic drugs. These engineered antibodies are activated at key “checkpoints” to counter cancer cell signalling (“check-point inhibitor”) and thus help the body of the host to 1) find and 2) destroy cancer cells that are reliant on these evolutionary survival pathways.
In 2001 Gleevec (Imatinib), was one of the first drugs to make a splash. Imatinib is a Tyrosine Kinase inhibitor that showed outstanding results in CML-type cancers and other types of cancers. This breakthrough kickstarted the rise to fame of this class of drugs. Later in 2014, the FDA approved Pembrolizumab (PD-1 Ligand) for use in melanoma and later many other solid tumors.
These two drugs were extremely impactful developments in the field and have saved many lives. But since then, a lot of other drugs in this class haven’t delivered on their early-phase trial results. The immune system is extremely complicated and not fully understood in terms of basic science. Therefore, a lot of cancers have had no new immunotherapy advances in the past 20 years and for those instances where there are new drugs, we’ve often seen inconsistent results i.e. many non-responders.
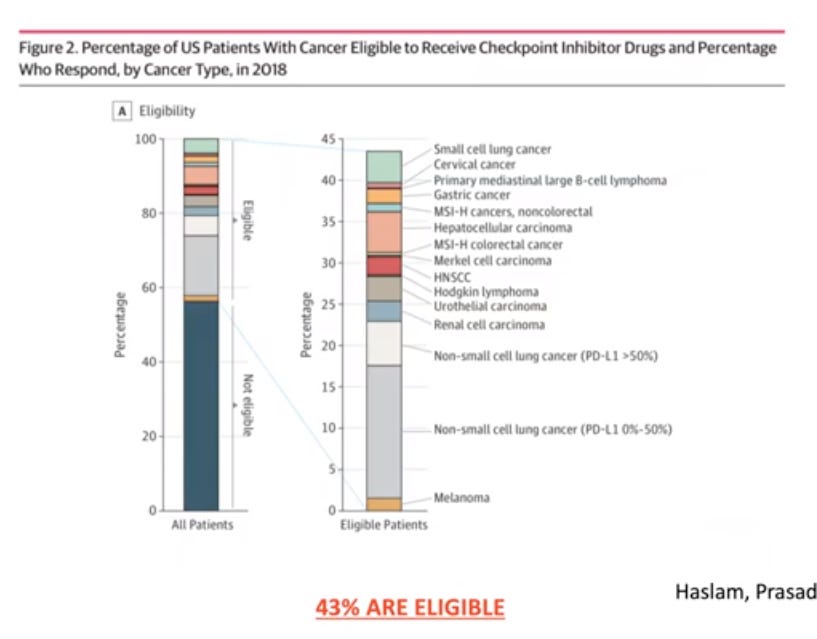
As you can see here, only about 43% of all solid cancer patients are eligible for an immunotherapy drug.
In terms of median responsiveness (i.e. complete response), consensus is that only a fraction of eligible patients will see any benefit from starting immunotherapy. The reasons as to why this is the case are not fully understood but the leading theory is that the degree of target marker expression and number of mutations are two important distinctions as to which patients have a lasting response.
This is illustrated well in this table, the highest response rate is around 50% in cutaneous squamous cell. For pancreatic cancer as you can see, not much has made a lasting impact within this class of drugs. Still not good enough.
Other undeperformers: Genome-based therapies
To start us off, here is a graph of genomic testing costs (hint: they have plummeted).
The smaller graph below the cost-analysis is a plot showing the rate of development of genome-targeted therapies. As you can see, it is not an explosion to match the fall in research costs, but more of a steady uptick. This means that patients are not seeing any big differences in outcomes from this class of therapies.
Many new genomic “targets” are being discovered but why hasn’t that led to extensive drug discovery and survival?
Well, some of that can be explained by basic economics.
Drug development is an expensive, long, and uncertain process. As a profit-driven mechanism, it is logical that old targets that have shown some promise are prioritized in clinical trials over newer genomic targets that might not lead to improved outcomes over currently available treatments.
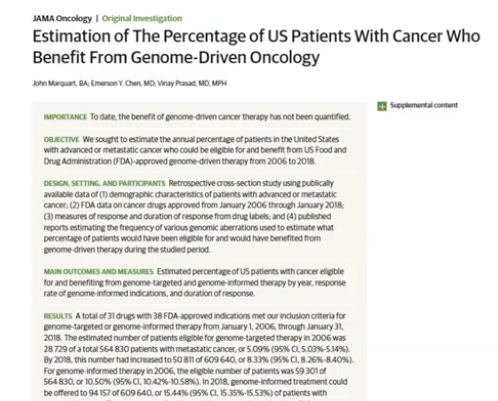
In 2018, a large study published in JAMA oncology found that only about 8% of patients were standing to gain any benefit from genome-driven therapies (small percentage of a small percentage= small market).
This low percentage is because many cancers have mutations that are not considered to be “driver-mutations” and so targeting these mutations will not have long-lasting clinical benefit because they are not crucial to the cancer’s survival mechanisms.
Regardless of your thoughts on whether only 8% of patients truly would benefit or the ethical implications of these failures. The reality of this statistic suggests that most patients have not benefited so far from the advances in genomics. It does play a role in the care of some patients, especially diagnostic (genetic counselling, screening decisions, prognostication) but in terms of treatments, most patients are still using the 3 main modalitites: chemotherapy, surgery, and radiation.
Part 3. The Moonshots (to end on a positive note)
1. Imatinib (target: tyrosike kinase)
This is an incredible graph. When you hear of moonshots, think of Imatinib.
Imatinib is the brain-child of researchers at Novartis, it was introduced to the market in 2001, and things completely changed for CML patients. It is a monoclonal antibody built specifically to target cells overexpressing the tyrosine kinase gene. The effects on life expectancy have been staggering, in fact looking at (2000-2022), the life expectancy gap has closed. This is the ideal situation for any cancer therapeutic and hopefully there will be many more to come.
Closing thoughts
Thank you for taking the time to read. I shared a lot of information here to hopefully paint a clear picture of the overall state of drug discovery in oncology. Looking at drugs like imatinib, I am incredibly optimistic about the future of cancer medicine. But I also remain wary of how little we can do for stage 4 cancer patients within our current paradigm.
Hopefully, this nuance is something that you will also come away with after reading this essay. In the unfortunate event that you or a loved one experiences this disease,
I wish you continued strength and dignity.
We will close with one of my favorite poems by the great Emily Dickinson.
Because I could not stop for Death – He kindly stopped for me – The Carriage held but just Ourselves – And Immortality.
We slowly drove – He knew no haste And I had put away My labor and my leisure too, For His Civility –
We passed the School, where Children strove At Recess – in the Ring – We passed the Fields of Gazing Grain – We passed the Setting Sun –
Or rather – He passed Us – The Dews drew quivering and Chill – For only Gossamer, my Gown – My Tippet – only Tulle –
We paused before a House that seemed A Swelling of the Ground – The Roof was scarcely visible – The Cornice – in the Ground –
Since then – 'tis Centuries – and yet Feels shorter than the Day I first surmised the Horses' Heads Were toward Eternity –
—1
References
Marquart J, Chen EY, Prasad V. Estimation of the Percentage of US Patients With Cancer Who Benefit From Genome-Driven Oncology. JAMA Oncol. 2018;4(8):1093–1098. doi:10.1001/jamaoncol.2018.1660
Malignant: How Bad Policy and Bad Evidence Harm People with Cancer. Vinay Prasad, published in 2020 by Johns Hopkins University Press.[1]